Biochemical assays in Hit Characterization
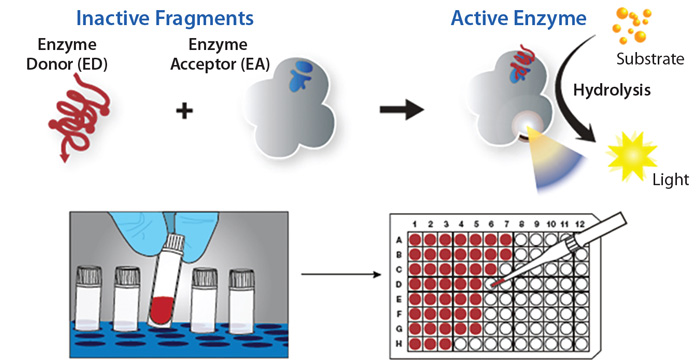
Fig.1 One of the biochemical assays: enzymatic assays. (Neil, W. C.; et al. 2016)
Biochemical assays are a set of approaches performed for detecting, quantifying and studying the activity of biological substances. Diverse read out techniques including ELISA, FRET and Western blotting are applied for quantification of metabolic activity. Functional behavior of multiple biomolecules including enzymes, proteins and small molecules can be measured utilizing biochemical assays. In addition, biochemical technologies are also able to identify protein-protein and protein-DNA/RNA interaction.
Our Services of Biochemical Assays
Our biochemical teams have comprehensive capabilities in development and performance of functional biochemical assays, including enzymatic assays, receptor ligand-binding assays, etc.
Enzymatic Assays
- Evaluate enzyme kinetics by measuring enzyme parameters such as kcat and Km.
- Study mechanism of action and enzymatic assays (determination of inhibitor on/off rates, Ki and kinact/KI for slow off-rate or covalent binders).
- Monitor enzyme purity and identity to ensure its specific activities.
Binding Assays
- Optimize the binding condition including investigating the reagent specificity, binding affinity and specific activity to ensure the assay design is fit-for-purpose.
- Minimize matrix interference, cross-reactivity, or nonspecific interactions in a complex biological matrix throughout the life of your program.
- Validate the mechanism of action and in vitro function.
Higher throughput screening
We have developed and optimized microplate-based screening and make our biochemical assays capable of higher throughput and more robust.
- Assay miniaturization: Miniaturization of screening assays to high-density microplate formats (384 or 1536 wells).
- Assay optimization: Including well-wide metrics such as Z’, robust Z’, dynamic range and signal to background.
- Calculate and track key QC measures (such as Z’ factor) automatically.
- Import data from our plate-readers directly.
Assay Format
BOC Sciences provides a full package of various assays with our comprehensive instrumentation and methodology, including:
- Spectrophotometry (absorbance, radiometry).
- Fluorescent (HTRF, FP, FRET, FI, AlphaLISA, AlphaScreen, Glo, Luciferase, calcium flux, etc.).
- Luminescent (ELISA, Alpha-tech, BRET, Nano-BRET, Glo assay, etc.).
- Gel-based assays (pulldown, WB, etc.).
- HPLC and LC/MS methodologies.
Advantages of Biochemical Assays Services
- Our biochemical teams can quickly establish optimum buffer conditions and continuously conduct the quality control of the assay outcome.
- We offer reliable data analysis with no manual intervention and full traceability of raw data.
- BOC Sciences also provides full customization of workflow and assay formats available to support drug discovery efforts in both small and large molecules.
- Here we present required and high-quality biochemical assays for comprehensive characterization of the target as well as helping you to understand its function.
Reference
- Neil, W. C.; et al. Accelerating Biologic and Biosimilar Drug DevelopmentReady-to-Use, Cell-Based Assays for Potency and Lot-Release Testing. BioProcess International. 2016, 14(1): 36-44.
※ It should be noted that our service is only used for research.

One-stop
Drug Discovery Services
- Experienced and qualified scientists functioning as project managers or study director
- Independent quality unit assuring regulatory compliance
- Methods validated per ICH GLP/GMP guidelines
- Rigorous sample tracking and handling procedures to prevent mistakes
- Controlled laboratory environment to prevent a whole new level of success
Online Inquiry

