Phenotypic Assays
A phenotype is the collection of observable traits of an organism including size, shape or molecular content. Phenotype assay is a cellular-based assay monitoring phenotype changes of relevant disease models and enables to simultaneously collect and quantify a large amount of multi-parametric data and information such as distribution patterns of the receptor, how spread out the cells are, and cellular morphology. The biggest advantage of phenotype assay is its capability of testing a host of compounds in a target agnostic assay that monitors phenotypic changes. Phenotypic drug discovery with high content screening therefore has developed an indispensable tool in hit identification.
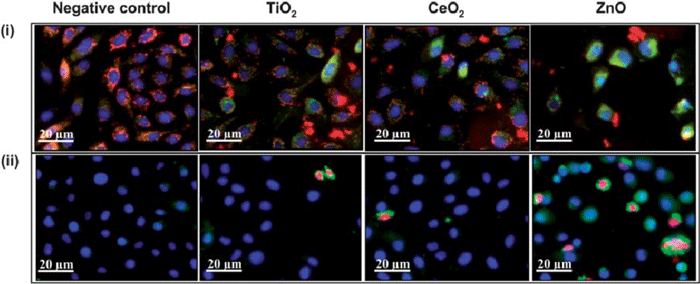 Fig.1 Phenotypic assays for monitoring cytotoxic events triggered by nanoparticle interaction in which healthy cells show blue nuclei, while damaged cells show green cytoplasm and red nuclei. (Damoiseaux, R.; et al. 2011)
Fig.1 Phenotypic assays for monitoring cytotoxic events triggered by nanoparticle interaction in which healthy cells show blue nuclei, while damaged cells show green cytoplasm and red nuclei. (Damoiseaux, R.; et al. 2011)Services of Phenotypic Assays
BOC Sciences has rich experience in conducting phenotypic assays and we provide the following detailed services in our phenotype assays:
Assay development
Our teams provide assay development services of selecting the right cell type and assay parameters of phenotypic assays.
1. Cell types
Patient-derived cells (healthy/diseased).
Stem cells and tumor cell lines.
Human primary cells.
Genome engineered cells (CRISPR/Cas9 gene editing).
2. Read-out
Fluorescence-based.
Luminescence-based.
Mass-spectrometry-based.
Compounds libraries screening
BOC Sciences offers target-focused phenotypic screening libraries and several other compound collections for your high content screening. We have also developed assays for small molecule screening using our broad compound collections and for RNAi screening using our adenoviral platform. Our team provides customized libraries to meet your project requirements either.
Biomarker Identification
We provide a well-validated collection of immunologically diverse human peripheral blood mononuclear cells to assess the effect of drug candidates on immune-related protein biomarkers. Our optimized assays enable to accommodate a wide range of biologics and small molecules with the application of flexible readouts and time-point format.
Phenotypic changes monitoring
We can monitor a series of phenotypic changes (including cellular morphology, sub-cellular localization, locations of co-staining, presence/absence of certain cell types, clustering in certain regions, etc.) and deliver bioactive compounds based on these disease-relevant parameters. Both visualization and quantification in real time are available in our phenotypic assays experiments.
Target accessibility evaluation
Our experts provide the assessment of target accessibility by monitoring and evaluating whether the compounds cross the cell wall or membrane and have intracellular interactions with the target successfully.
In vitro toxicity studies
We perform the cytotoxicity investigation by using the obtained phenotypic parameters associated with toxicity in a single, complex biological system, such as viability, hypoxia, translocation, oxidative stress, mitochondrial dysfunction, etc.
Advantages of Phenotypic Assays Services
- With professional knowledge in cellomics and rich experiences in cell biology experiments, our scientists can conduct multiplexing assays to identify compounds with the ability to treat diseases.
- We provide predictive and translatable data obtained from phenotypic assays for your further vivo studies.
Reference
- Damoiseaux, R.; et al. No time to lose-high throughput screening to assess nanomaterial safety. Nanoscale. 2011, 3: 1345-1360.
※ It should be noted that our service is only used for research.

One-stop
Drug Discovery Services
- Experienced and qualified scientists functioning as project managers or study director
- Independent quality unit assuring regulatory compliance
- Methods validated per ICH GLP/GMP guidelines
- Rigorous sample tracking and handling procedures to prevent mistakes
- Controlled laboratory environment to prevent a whole new level of success
Online Inquiry

