Biophysical Assays in Hit Characterization
Biophysical methods monitor and measure binding events between two molecules, such as two proteins or a protein and a small molecule. The information obtained can be utilized for establishing structure-activity relationships in target identification and validation. Nowadays, as indispensable tools, biophysical methods have been widely used in different stages of drug development from primary screening to lead optimization.
Fig.1 The Picture of Biophysical assays. (Tatum, N. J.; et al., 2017)
Capabilities of biophysical assays
As affinity based screening and characterization tools, each biophysical method can maximize the chances of identifying binders to molecular targets and avoid the waste of resources as possible. In combination, these broad panel of techniques can provide vital affinity, kinetic, thermodynamic and structural data on ligands binding to their targets.
- Vital kinetic data on ligand binding to its target can be obtained by Thermal Shift Assays(TSA), Surface Plasmon Resonance (SPR) spectrometry.
- Isothermal Titration Calorimetry (ITC) and Thermal Shift Assays (TSA)study thermodynamic information about protein-ligand interactions.
- Further analysis of mode of action studies and protein-protein / protein-ligand interactions are provided by Surface Plasmon Resonance (SPR), high-field Nuclear Magnetic Resonance(NMR), Microscale Thermophoresis (MST) and Mass Spectrometry(MS)。
- Affinity study can be done by Isothermal Titration Calorimetry (ITC).
- Circular Dichroism (CD) spectroscopy determines the protein structure and assesses its physical stability.
Advantages of Biophysical Assays
1.Allow label-free detection
Biophysical measurement corresponds to a biophysical variation directly.
2.Biophysical Analysis
Biophysical Assays provide not only an orthogonal measurement but also offer additional parameters, such as thermodynamic, kinetics, affinity and structural information.
Biophysical assay in HTS
Biophysical assay technique has become an established approach in HTS hit triaging, owing to its high quality in detecting protein-ligand interactions. Information obtained from biophysical analysis are useful in quality control of protein and works in following three aspects:
To assist design of HTS assays
To confirm binding of HTS hits
To guide structural biology drug discovery effort.
More specifically, biophysical technology plays a role in both pre-HTS and post-HTS to improve the success rate in hit-finding campaigns:
- In the pre-HTS stage, biophysical techniques assist in developing a robust and suitable screeing assay.
- In the post-HTS stage, biophysical assay improve the efficient by filtering HTS output based on characterization potency in hits finding campaigns.
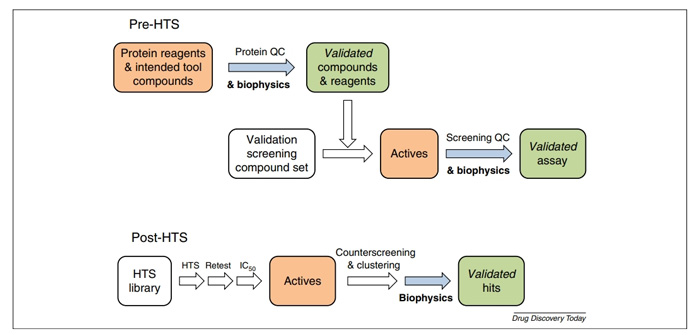
Fig.2 In tegration of biophysical assays in HTS-driven hit-finding projects. (Folmer, Rutger, H. A, 2016)
As important part of our ongoing efforts to provide our clients with high-quality services to support their drug discovery processes, BOC Sciences introduces our Biophysical assay services:
Surface Plasmon Resonance (SPR) Spectrometry
Surface plasmon resonance spectroscopy is a rapidly developing sensitive technique for the study of ligand binding interactions with membrane proteins, which are the major molecular targets. It’s capable of monitoring the binding between two molecules in real time and screening tens of thousands of compounds in a very short time. We can perform an orthogonal binding analysis using SPR spectrometry to eliminate false positives in your drug discovery project.
Thermal Shift Assay (TSA)
Thermal shift assay is one of the label-free technologies and has gained popularity in drug discovery. It measures the thermal stability of a target protein and the increase in protein melting temperature(Tm) upon the binding of a stabilizing agent. In recent years, TSA has developed a useful tool for identifying ligands, buffer conditions, and co-factors affecting protein stability and are compatible with high-throughput screening. Our TSA group can offer custom-tailored thermal stability assay development for many kind of target categories to meet your specific need.
Microscale Thermophoresis (MST)
As an emerging and sensitive method, Microscale Thermophoresis assays (MST assays) is applied to study the interaction between molecules, such as proteins and small molecules. It measures the motion of molecules along temperature gradients that changes upon ligand binding and allows for quantification of the binding and determination of affinity. Our scientists utilize MST to conduct analysis of biomolecules interaction and triage the output of HTS campaigns, helping to improve the efficiency of drug discovery.
Isothermal Titration Calorimetry (ITC)
Isothermal titration calorimetry is a relative new technology based on the measurement of the heat released / absorbed during the binding event of a compound to its ligand. Not only it offers the information of binding affinity through the data of binding paramaters obtained but also the mechanisms of potential molecular interactions. With the application of ITC method, we provide help to make right decisions about lead compound selection and optimization with deeper understanding and profound insight of structural-functional relationships.
Nuclear Magnetic Resonance (NMR)
As a powerful screening and analytical tool, NMR is applied to screen chemical fragment libraries in the new drug development and remains the preferred technique for determining the molecular structure. NMR provides many useful parameters, such as chemical shift, diffusion coefficient, transverse relaxation, NOE, etc., which are crucial for the selection of ligand compounds. Another important characteristic is that NMR can reveal the structural information related to the ligand-protein interaction and serves as reference for designing ligand. Scientists from BOC Sciences study the interaction between the ligand and the target, providing structural information obtained from the NMR experiments for the validation of hit and design of lead compounds.
Circular Dichroism (CD) Spectroscopy
In recent years, Circular dichroism spectroscopy serves as a powerful tool for rapid determination of protein structure and assessing the physical stability of proteins. Compared to conventional techniques, CD spectroscopy only requires a minimal amount of sample. Complementary information about the secondary and tertiary structural state of a protein can also be collected for protein conformation from CD technique. BOC Sciences has developed a fully automated high throughput CD system for diverse applications including proteins structure studies, thermodynamics and protein-ligand interactions investigation and so on.
Mass Spectrometry (MS)
Mass spectrometry is a detection method by separating and analyzing samples in an ionized state. As an emerging label-free screening technology, MS has developed to be applied in a diversity of applications ranging from identifying metabolites and proteins to studying microbial interactions. In BOC Sciences, we have established multiple MS detection platforms combine with other separation technologies including Liquid Chromatography (LC) system, Capillary Electrophoresis (CE), Gas Chromatography (GC), etc. to fulfill different demand of our clients’ drug development plans.
Bio-Layer Interferometry (BLI)
Bio-Layer Interferometry is an biosensor based optical technique detecting the molecular complexes on its sensor-tip which have specific binding to one interaction partner. The advanced approach offers measurement of binding kinetics and affinity of biological macromolecule interactions in real time. Therefore, it is considered as a important tool for fast and reliable hit screening and validation based on affinity. At BOC Sciences, we have developed a high sensitivity and high throughput BLI assay capable of quantifying and determining the kinetics of biomolecule interactions. Our teams are confident in improving the speed and efficiency of your drug discovery process with our high-quality BLI services.
Our Capabilities of Biophysical Assays Services
- We have leading experts in handling both affinity and kinetic data and we use these knowledge to support each phase of your drug research projects(from early stage of hit finding to mechanistic profiling through lead optimization)
- BOC Sciences have established a comprehensive biophysical assay platform to obtain diverse data and provide accurate, real-time and label-free interaction analysis information to meet your demand.
※ It should be noted that our service is only used for research.

One-stop
Drug Discovery Services
- Experienced and qualified scientists functioning as project managers or study director
- Independent quality unit assuring regulatory compliance
- Methods validated per ICH GLP/GMP guidelines
- Rigorous sample tracking and handling procedures to prevent mistakes
- Controlled laboratory environment to prevent a whole new level of success
Online Inquiry

