Drug Repurposing
What is Drug Repurposing?
Drug repurposing or drug repositioning is a well-known approach applied by scientists to find new medical uses in the existing licensed or currently marketed drugs. Since huge amounts of research data enable to be re-analyzed with the application of informatics solutions, drug repurposing method is able to mitigate failures in drug development programs and accelerate the process.
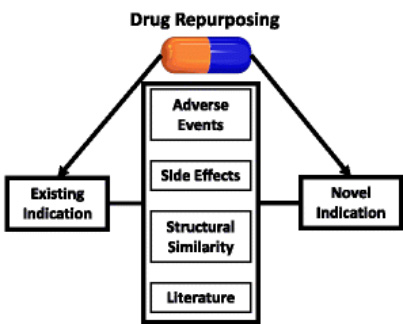
Fig.1 Systematic of drug repurposing process. (Ferguson, L. B.; et al. 2018)
Advantages of Drugs Repurposing
- Cost-effective: Significantly reduce research costs.
- Time-saving: Reduce time waste since the safety and efficacy of the existing drugs have been proved.
- Low risk: Reduce risk of unexpected side effects by repurposing drugs the existing drugs.reduce the risks of failure.
Drug Repurposing Services
Drug repurposing libraries screening
We have developed drug repurposing libraries for detecting candidates from thousands of drugs and bioactive molecules. All of the approved drugs at BOC Sciences are validated by NMR and HPLC and most of them are available for HTS. We also offer detailed compound information such as their structure and activity for your new drug target screening.
Identification of new drug repurposing
- Drug-protein relationships studies
We provide valuable information on drug-protein relationships according to our database such as targets, enzymes, carriers and transporters. In addition, off-target relationships, annotations describing pharmacological action and types of interaction (antagonist, agonist, substrate, inhibitor, and inducer) are also available for identifying drug repurposing candidates rapidly. - Indications studies
In order to improve the screening outcomes, our experts offer a variety of indications data. At BOC Sciences, comprehensive databases are available including approved indications, most common off-label indications and clinical trial indications. We use them to repurpose failed clinical trials and facilitate data integration, finding potential efficacy of drug candidates in more indications. - Side effects studies
Our scientists have collected information of the side effects from clinical trial data, drug labels and post-market reporting. We use them to help our customers to compare side effects and identify potential drug candidates by discovering drugs with similar off-target effects. - Pathway and metabolism studies
We share our resources of pathways and metabolic reactions databases with our clients to help them better understand downstream effects. We offer structural descriptions of metabolism and detailed disease pathways which are crucial in identification of the potential mechanisms. - Evaluation of the safety and effectiveness Toxicity investigation: We provide detailed toxicity data of a large number of market drugs to identify the potential toxicity of candidates drugs with similar structure, giving guidance on the structural modification.
- Drug-drug interactions studies: Accurate and updated information of drug-drug interactions are available at BOC Sciences for you to develop new drugs discovery projects effectively.
Our Advantages of Drug Repurposing Services
- Our drug repurposing libraries are continuously updated in order to offer comprehensive and up-to-date collections. Moreover, identity and purity of each drug have been experimentally confirmed.
- We can perform drug detection campaigns from thousands of medications and bioactive molecules with the application of high throughput technologies.
Reference
- Ferguson, L. B.; et al. From gene networks to drugs: systems pharmacology approaches for AUD. Psychopharmacology. 2018
※ It should be noted that our service is only used for research.

One-stop
Drug Discovery Services
- Experienced and qualified scientists functioning as project managers or study director
- Independent quality unit assuring regulatory compliance
- Methods validated per ICH GLP/GMP guidelines
- Rigorous sample tracking and handling procedures to prevent mistakes
- Controlled laboratory environment to prevent a whole new level of success
Online Inquiry

